Matthew Eroglu and his research team at the University of Toronto have discovered a new inheritance mechanism that operates completely independent of DNA or RNA. The team was first researching cancer signaling pathways using studying cancer signaling pathways using Caenorhabditis elegans, a species of worm used in genetic research. Instead of following the expected reproductive patterns, the worms had an unusual feminization over the following generations, becoming sterile after their research. Recognizing the anomaly, the team shifted their entire study to understanding the cause of this inheritance.
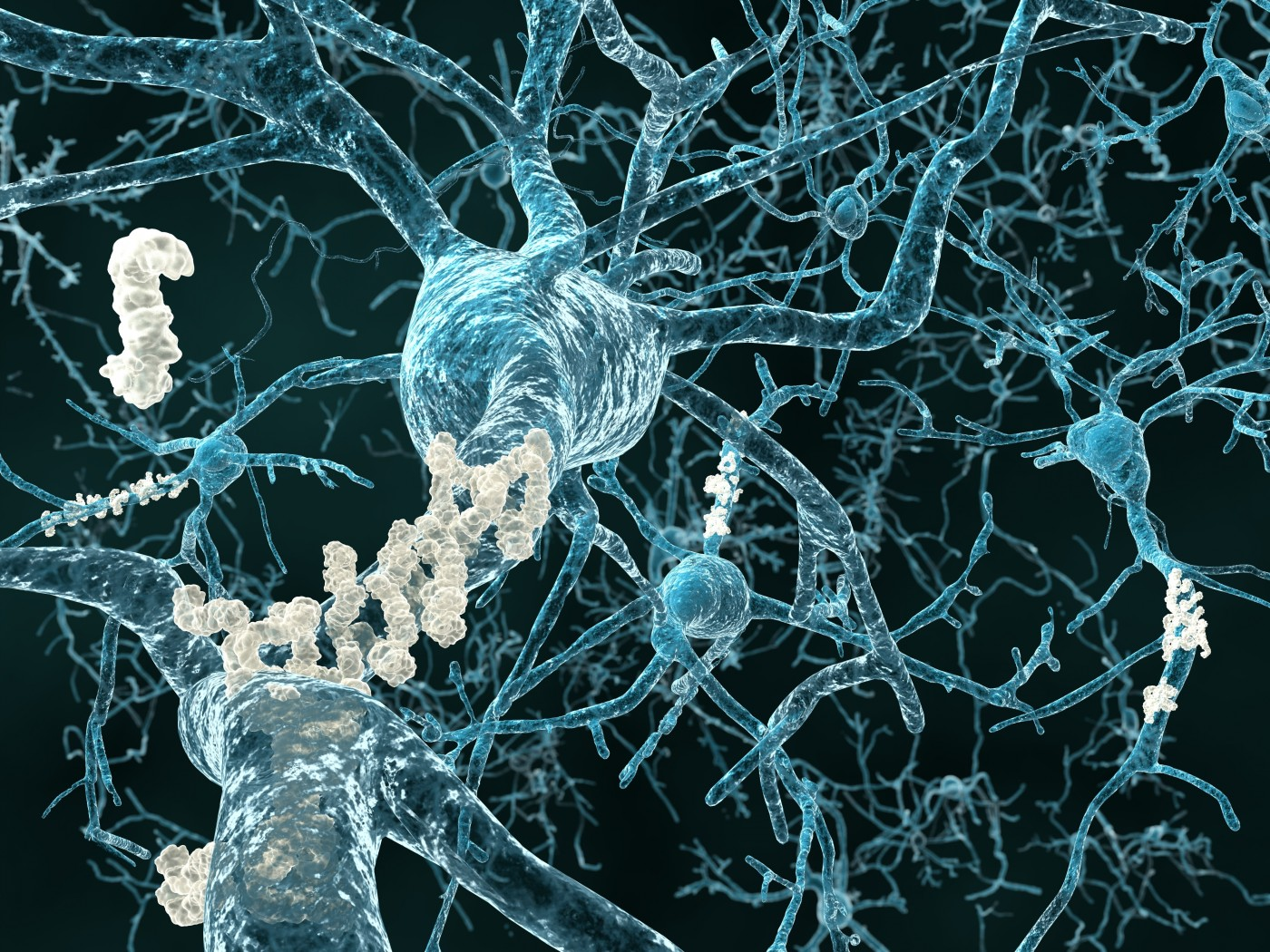
Their findings have changed our understanding of genetic inheritance forever, finding that the transmission of traits from one generation to the next could be done without genetic material, using proteins with amyloid structures and prion-like properties. This goes against our understanding of genetic inheritance, where DNA mutations or epigenetic modifications are the only way it changes. These amyloid proteins can self-replicate, influence cellular functions, and form microscopic aggregates, or “herasomes,” which accumulate over generations. As they accumulate, they only disrupt reproductive processes.
Amyloid proteins are typically associated with neurodegenerative diseases such as Alzheimer’s and Parkinson’s, where their ability to propagate in a prison-like manner allows for disease progression. However, in the case of C. elegans, these proteins changed the expression of key reproductive proteins, which changed the balance between sperm and oocyte production. Normally, the worms produce a set number of sperm cells before shifting to oocyte production. The amyloid aggregates disrupted this balance, gradually reducing sperm production and making more oocytes, which made the worms infertile in the long-term. Even more surprisingly, the process was reversible under lower-temperature conditions, showing its non-genetic, yet inheritable, nature.
This discovery can help us understand the “missing heritability” problem or the difference between observable inherited traits and the genetic variations identified through DNA analysis. Many complex traits and diseases, such as diabetes, cancer, and neurological disorders, have strong hereditary patterns that cannot be explained by traditional genetic studies. The presence of amyloid-based inheritance allows us to look at another layer of heritability, bridging the gap. Though we have not yet confirmed this in humans, the identification of amyloid aggregates in human oocytes allows for the possibility of a similar mechanism operating in higher organisms. Finally, this discovery is important when considering evolution as well. In C. elegans, the feminization effect may be an adaptive strategy in response to environmental stress, helping outcrossing with male worms rather than self-fertilization. This increased genetic diversity could enhance survival in changing conditions.
While the study raises numerous questions, it also opens new avenues of research into alternative mechanisms of inheritance. If amyloid-based inheritance is found to play a role in mammals, it could revolutionize our understanding of hereditary diseases and potentially offer life-altering therapeutic targets.













Leave a Reply